Salts are build of positive and negative ions which are bound together by the force of attraction of their opposite charges. If energy needed to brake their ionic bounds is lower then the energy given off by an interaction of the ions with a solvent (ie. water), the salts will dissociate and interact with solvent and thus dissolve.
At first the salt dissociates quickly and reaction is generally going one way:
NaCl(s) 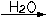 Na+(aq) + Cl-(aq)
Na+(aq) + Cl-(aq)
As more and more salt dissociates, the solution is getting more saturated. Eventually so many ions are dissolved in water, that a reverse reaction starts to take place:
Na+(aq) + Cl-(aq) 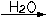 NaCl(s)
NaCl(s)
The reverse reaction decreases the speed at which water absorbes next particles of salt. Finally the rate of the reverse reaction reaches the rate of dissociation - the concentration of ions does not change with time. Solution reaches its equilibrium in given conditions and is called a saturated solution.
Solubility of salt is an amount of salt that needs to be added to water for the solution to reach an equilibrium.